ChemInform Abstract: The Chemistry of Transition Metals with Three-Membered Ring Heterocycles | Request PDF

Molecules | Free Full-Text | Modern Synthetic Methods for the Stereoselective Construction of 1,3-Dienes

Circularly Recyclable Polymers Featuring Topochemically Weakened Carbon–Carbon Bonds | Journal of the American Chemical Society
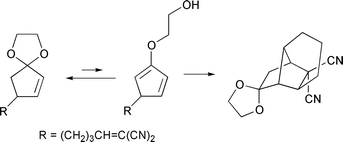
Intramolecular Diels–Alder reaction of cyclopenta-1,3-diene derivatives generated in situ from 4-(pent-4-enyl)cyclopent-2-enone ethylene ketals - Journal of the Chemical Society, Perkin Transactions 1 (RSC Publishing)

1Z,3E)-1,4-dimethoxy-1,3-butadiene - C6H10O2, density, melting point, boiling point, structural formula, synthesis
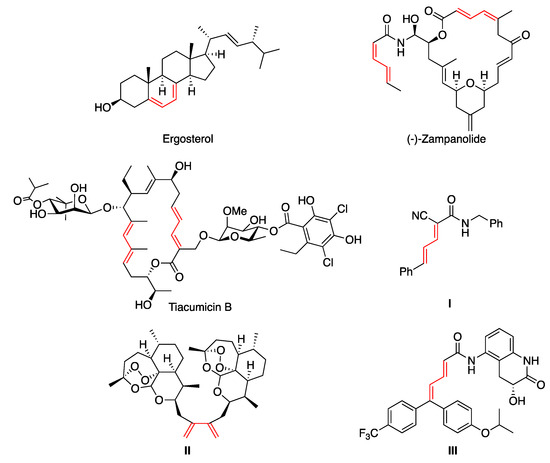
Molecules | Free Full-Text | Modern Synthetic Methods for the Stereoselective Construction of 1,3-Dienes

Diastereoselective Three-Component 3,4-Amino Oxygenation of 1,3-Dienes Catalyzed by a Cationic Heptamethylindenyl Rhodium(III) Complex | Journal of the American Chemical Society
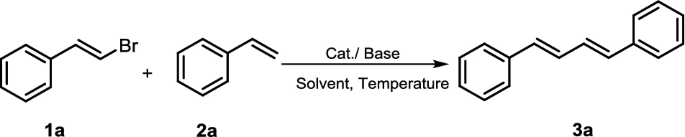
Synthesis of (1E,3E)-1,4-diarylbuta-1,3-dienes promoted by μ-OMs palladium–dimer complex | BMC Chemistry | Full Text

Palladium-Catalyzed Enantioselective Cycloaddition of Carbonylogous 1,4-Dipoles: Efficient Access to Chiral Cyclohexanones | Journal of the American Chemical Society
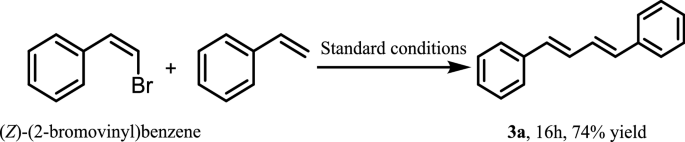
Synthesis of (1E,3E)-1,4-diarylbuta-1,3-dienes promoted by μ-OMs palladium–dimer complex | BMC Chemistry | Full Text
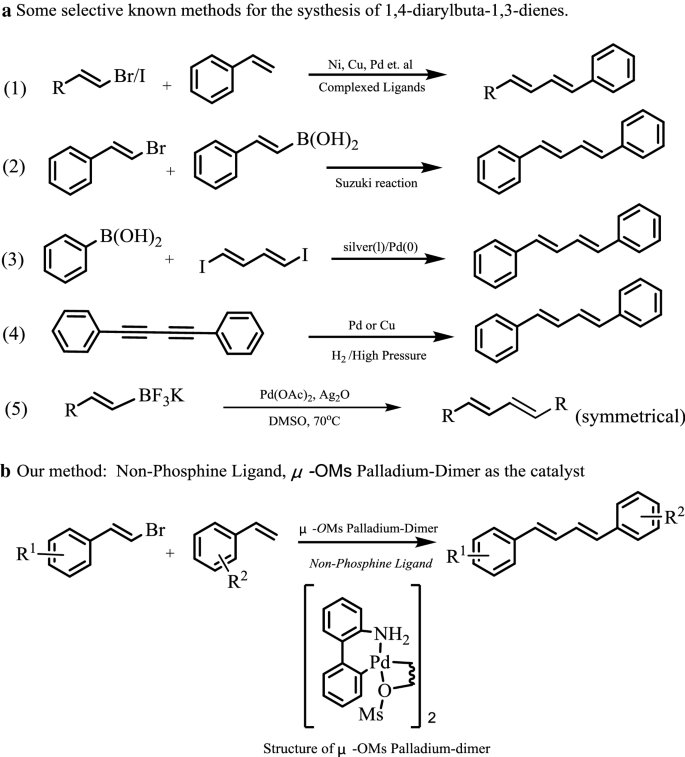
Synthesis of (1E,3E)-1,4-diarylbuta-1,3-dienes promoted by μ-OMs palladium–dimer complex | BMC Chemistry | Full Text
![Diastereo- and enantioselective palladium-catalyzed dearomative [4 + 2] cycloaddition of 3-nitroindoles - ScienceDirect Diastereo- and enantioselective palladium-catalyzed dearomative [4 + 2] cycloaddition of 3-nitroindoles - ScienceDirect](https://ars.els-cdn.com/content/image/1-s2.0-S1001841719301986-fx1.jpg)
Diastereo- and enantioselective palladium-catalyzed dearomative [4 + 2] cycloaddition of 3-nitroindoles - ScienceDirect