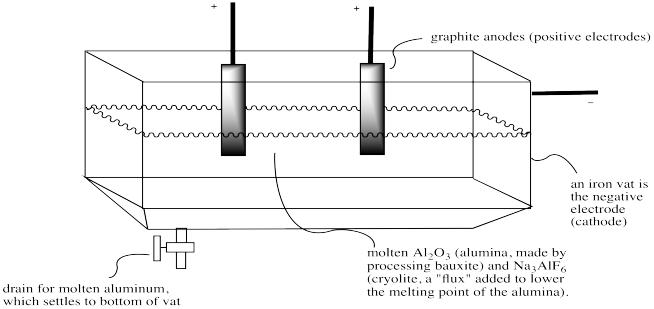
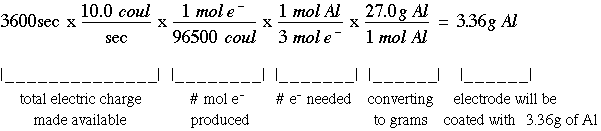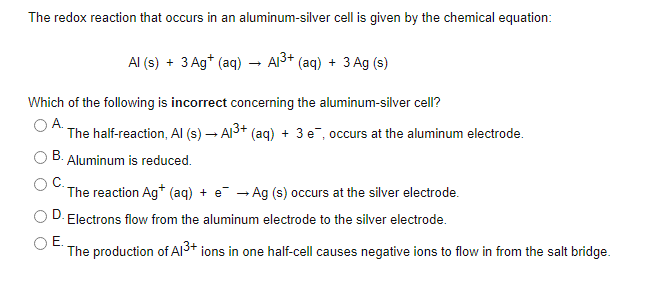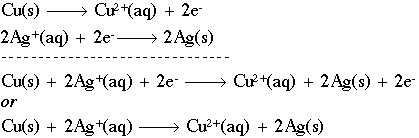Synthesis of Alum Lab This synthesis reaction involves a redox reaction and the formation of a complex ion. - ppt download

Recent Advances in Organic Reactions Involving Elemental Sulfur - Nguyen - 2017 - Advanced Synthesis & Catalysis - Wiley Online Library

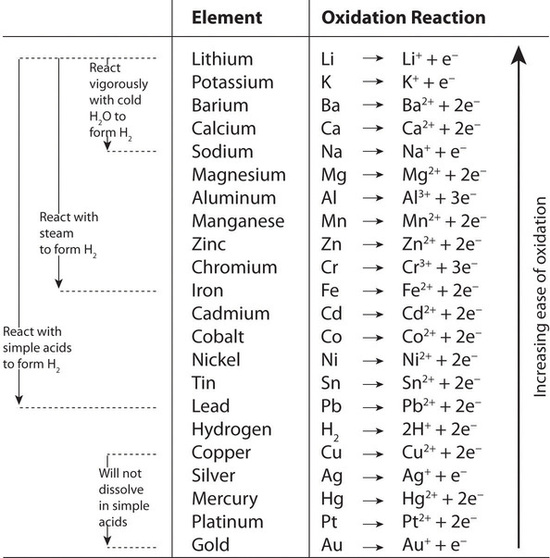
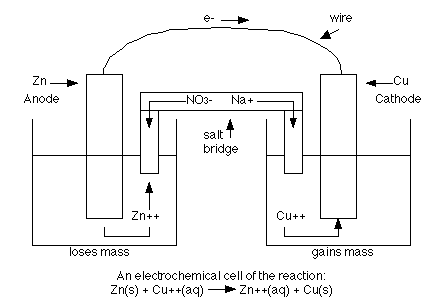
Redox Difficult but necessary. Obviously: Oxidation is adding oxygen 2H 2 + O 2 2H 2 O Reduction is removing oxygen 2FeO + C 2Fe + CO 2 But also oxidation. - ppt download

SOLVED: Aluminium metal is added to a solution of Cu(Il) ions, producing Aluminium ions and copper metal. The unbalanced redox equation for this reaction is below: a. Balance the oxidation half-equation. Al(s)
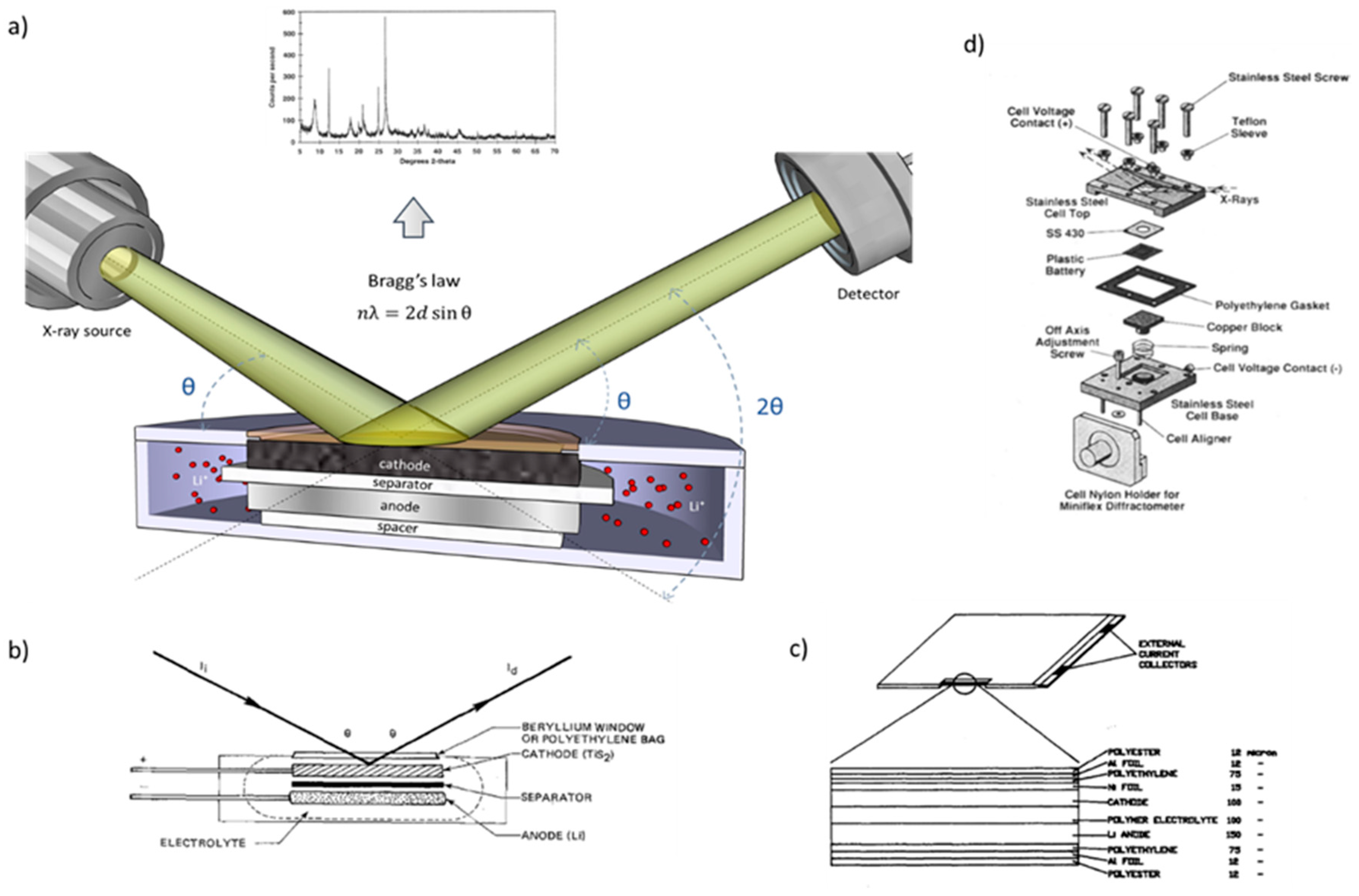
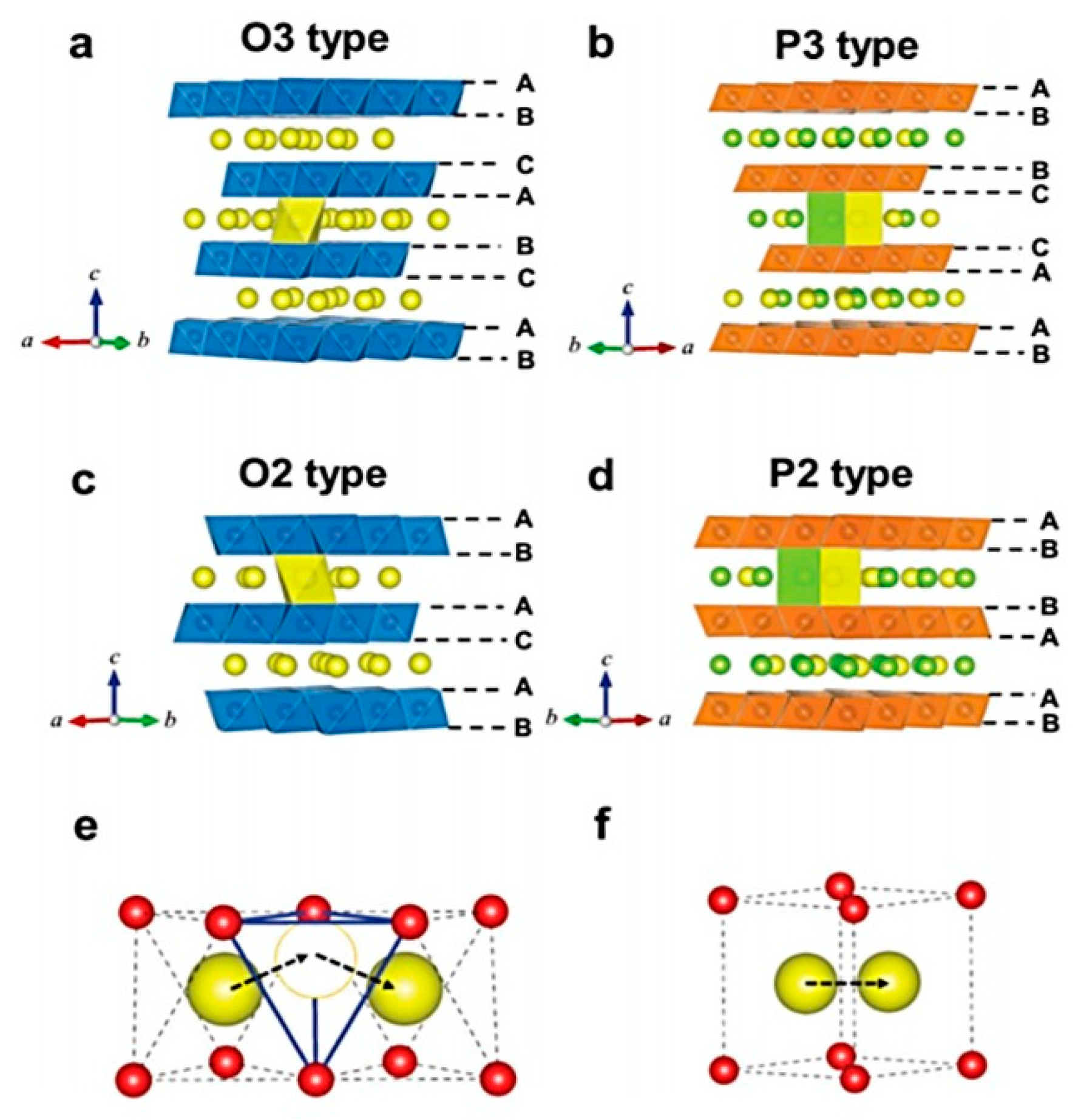
Condensed Matter | Free Full-Text | Using In-Situ Laboratory and Synchrotron-Based X-ray Diffraction for Lithium-Ion Batteries Characterization: A Review on Recent Developments

Recent Advances in Rechargeable Aluminum-Ion Batteries and Considerations for Their Future Progress | ACS Applied Energy Materials