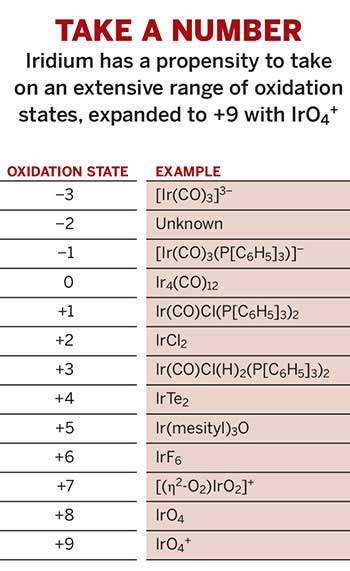
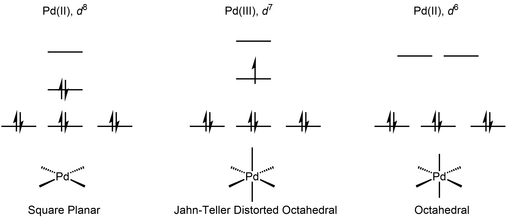
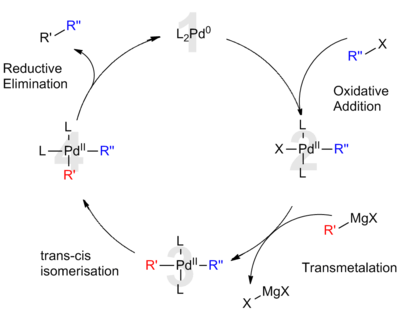
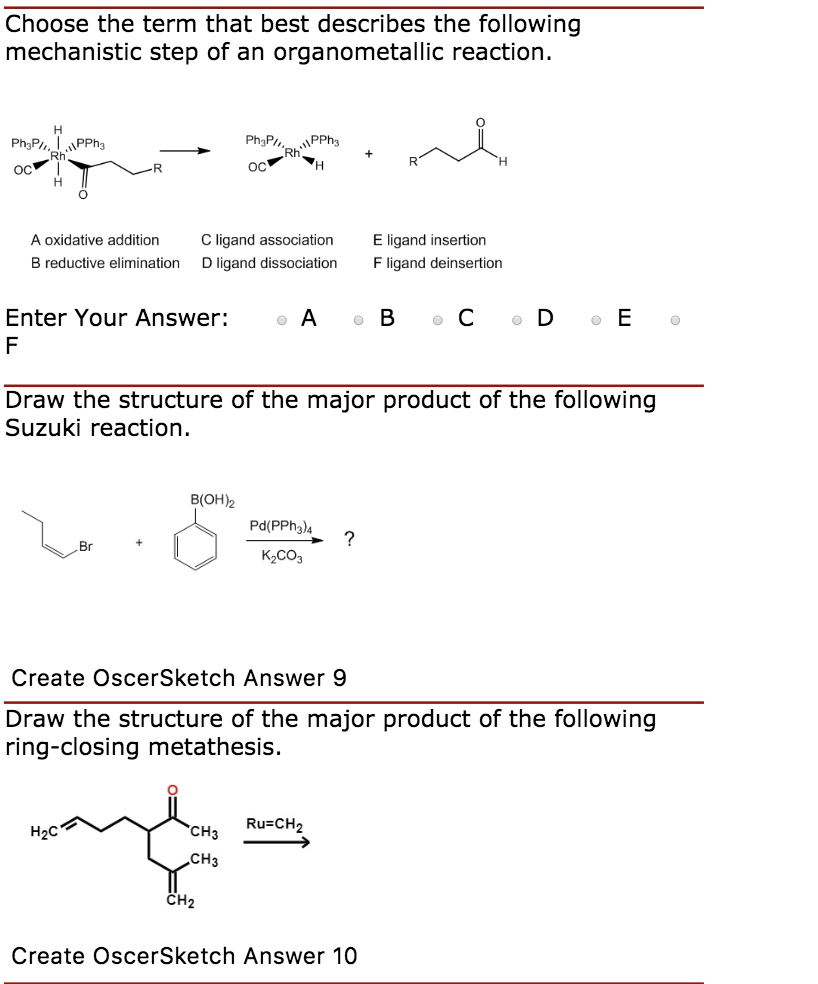
Table 1 from Ligand effects in bimetallic high oxidation state palladium systems. | Semantic Scholar
![PDF] Palladium(II)-Catalyzed Addition Reactions : Synthesis of Aryl Amidines and Aryl Ketones | Semantic Scholar PDF] Palladium(II)-Catalyzed Addition Reactions : Synthesis of Aryl Amidines and Aryl Ketones | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e8bb6604a650393f606af41e5bd4761322ed1e19/13-Table1-1.png)
PDF] Palladium(II)-Catalyzed Addition Reactions : Synthesis of Aryl Amidines and Aryl Ketones | Semantic Scholar
![PDF] In situ XAS study of the local structure and oxidation state evolution of palladium in a reduced graphene oxide supported Pd(ii) carbene complex during an undirected C–H acetoxylation reaction | Semantic PDF] In situ XAS study of the local structure and oxidation state evolution of palladium in a reduced graphene oxide supported Pd(ii) carbene complex during an undirected C–H acetoxylation reaction | Semantic](https://d3i71xaburhd42.cloudfront.net/84dfbbd87d26e94b879a52253b382bca49cb09f4/4-Figure1-1.png)
PDF] In situ XAS study of the local structure and oxidation state evolution of palladium in a reduced graphene oxide supported Pd(ii) carbene complex during an undirected C–H acetoxylation reaction | Semantic

Characterization and oxidation states of Cu and Pd in Pd–CuO/ZnO/ZrO2 catalysts for hydrogen production by methanol partial oxidation - ScienceDirect

High‐Oxidation‐State Palladium Catalysis: New Reactivity for Organic Synthesis - Muñiz - 2009 - Angewandte Chemie International Edition - Wiley Online Library

High‐Oxidation‐State Palladium Catalysis: New Reactivity for Organic Synthesis - Muñiz - 2009 - Angewandte Chemie International Edition - Wiley Online Library
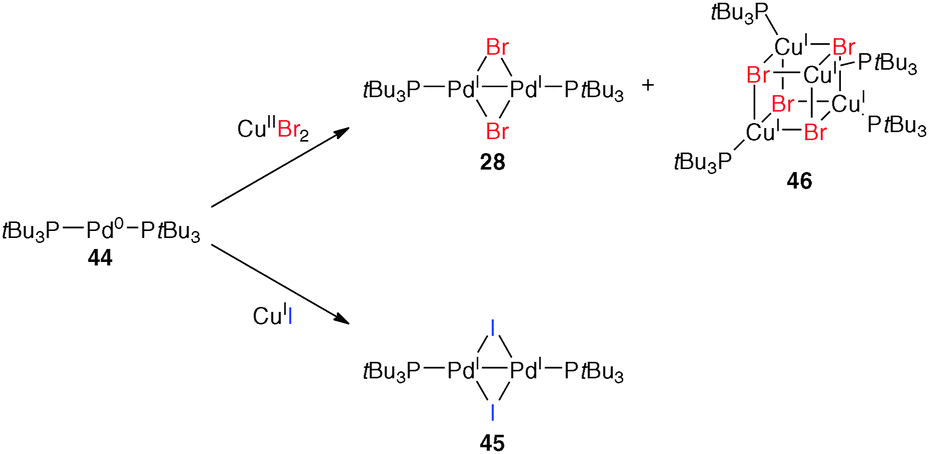
Experiment and computation: a combined approach to study the reactivity of palladium complexes in oxidation states 0 to iv - Chemical Society Reviews (RSC Publishing) DOI:10.1039/C4CS00061G
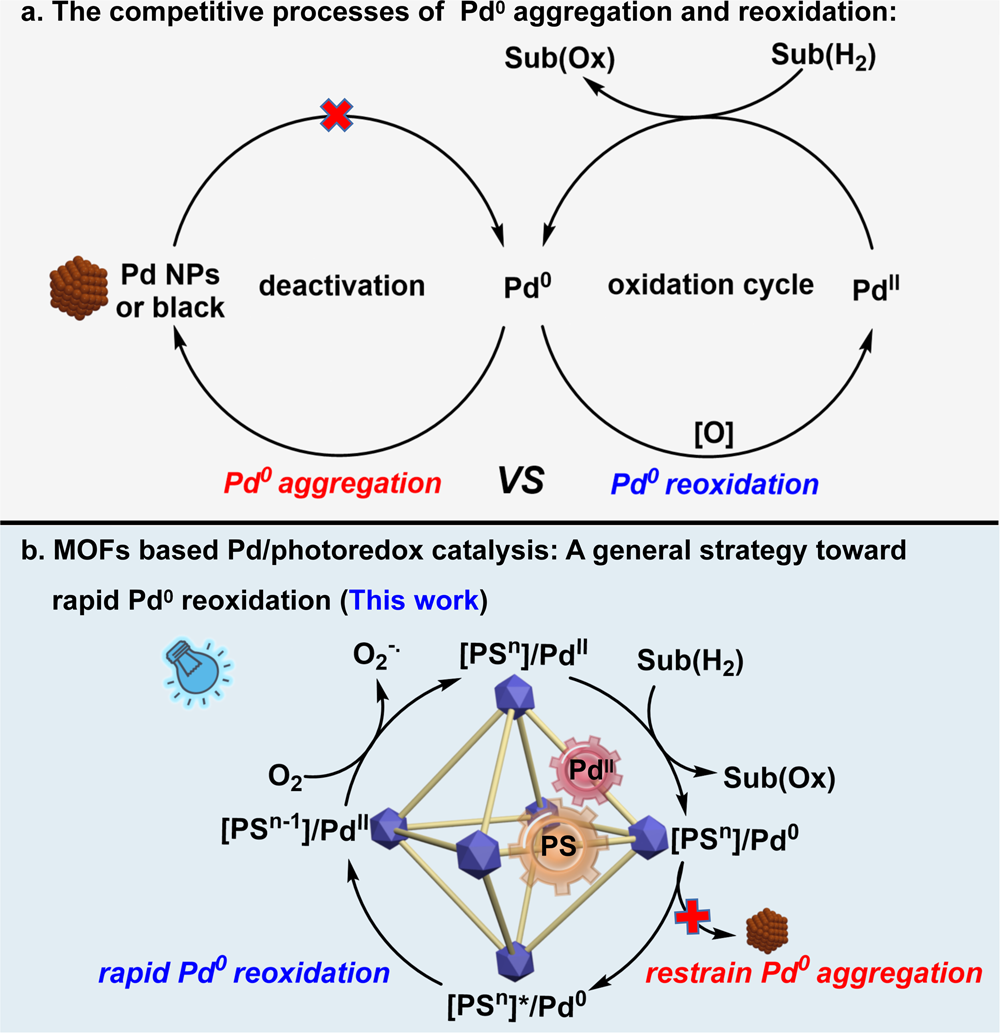
Visible light-driven efficient palladium catalyst turnover in oxidative transformations within confined frameworks | Nature Communications

Palladium Oxidase Catalysis: Selective Oxidation of Organic Chemicals by Direct Dioxygen‐Coupled Turnover - Stahl - 2004 - Angewandte Chemie International Edition - Wiley Online Library

Dual Oxidation State Tandem Catalysis in the Palladium-Catalyzed Isomerization of Alkynyl Epoxides to Furans | ACS Catalysis

How Does the Oxidation State of Palladium Surfaces Affect the Reactivity and Selectivity of Direct Synthesis of Hydrogen Peroxide from Hydrogen and Oxygen Gases? A Density Functional Study | Journal of the

The role of Pd–Pt Interactions in the Oxidation and Sulfur Resistance of Bimetallic Pd–Pt/γ-Al2O3 Diesel Oxidation Catalysts | Industrial & Engineering Chemistry Research