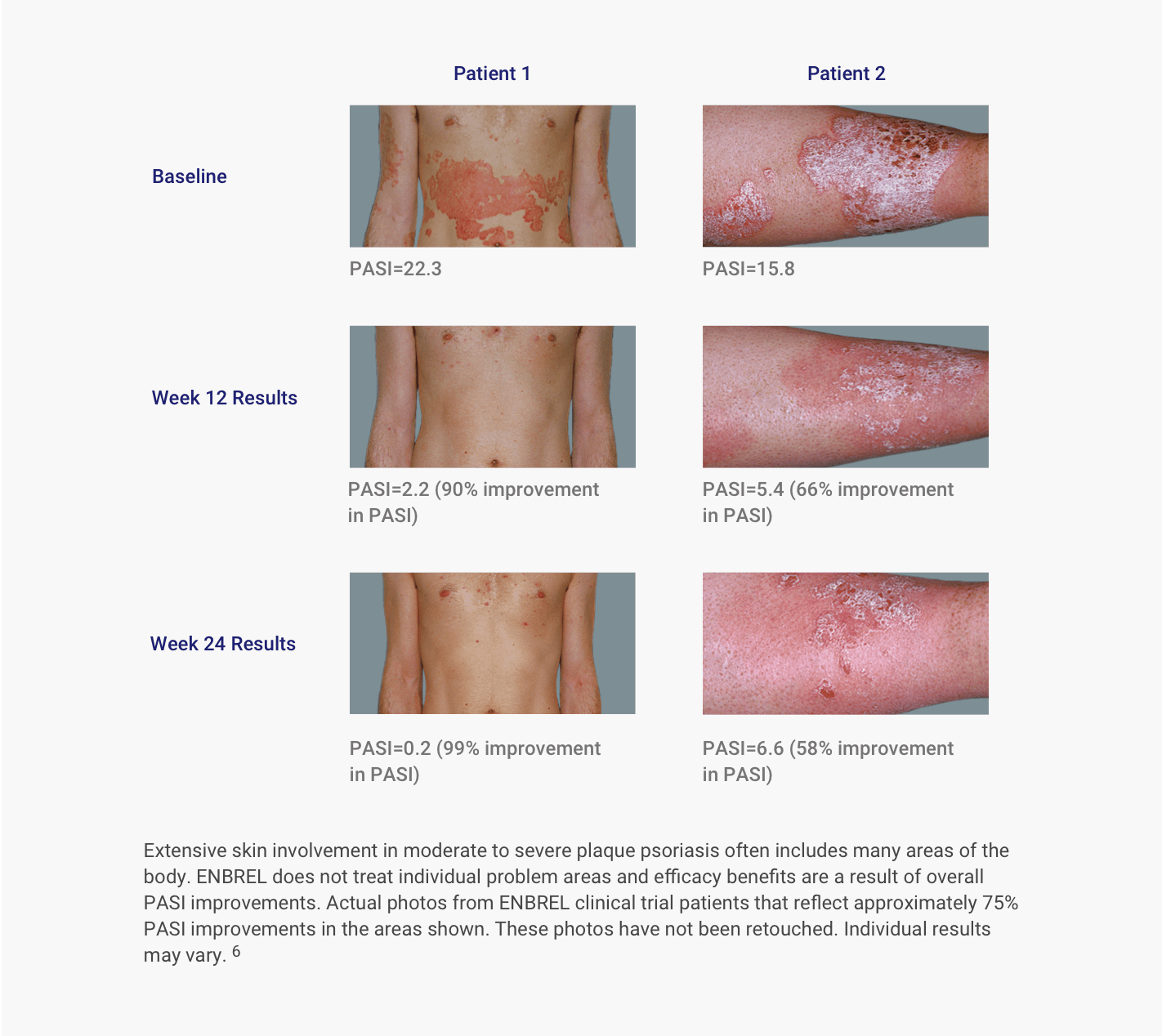
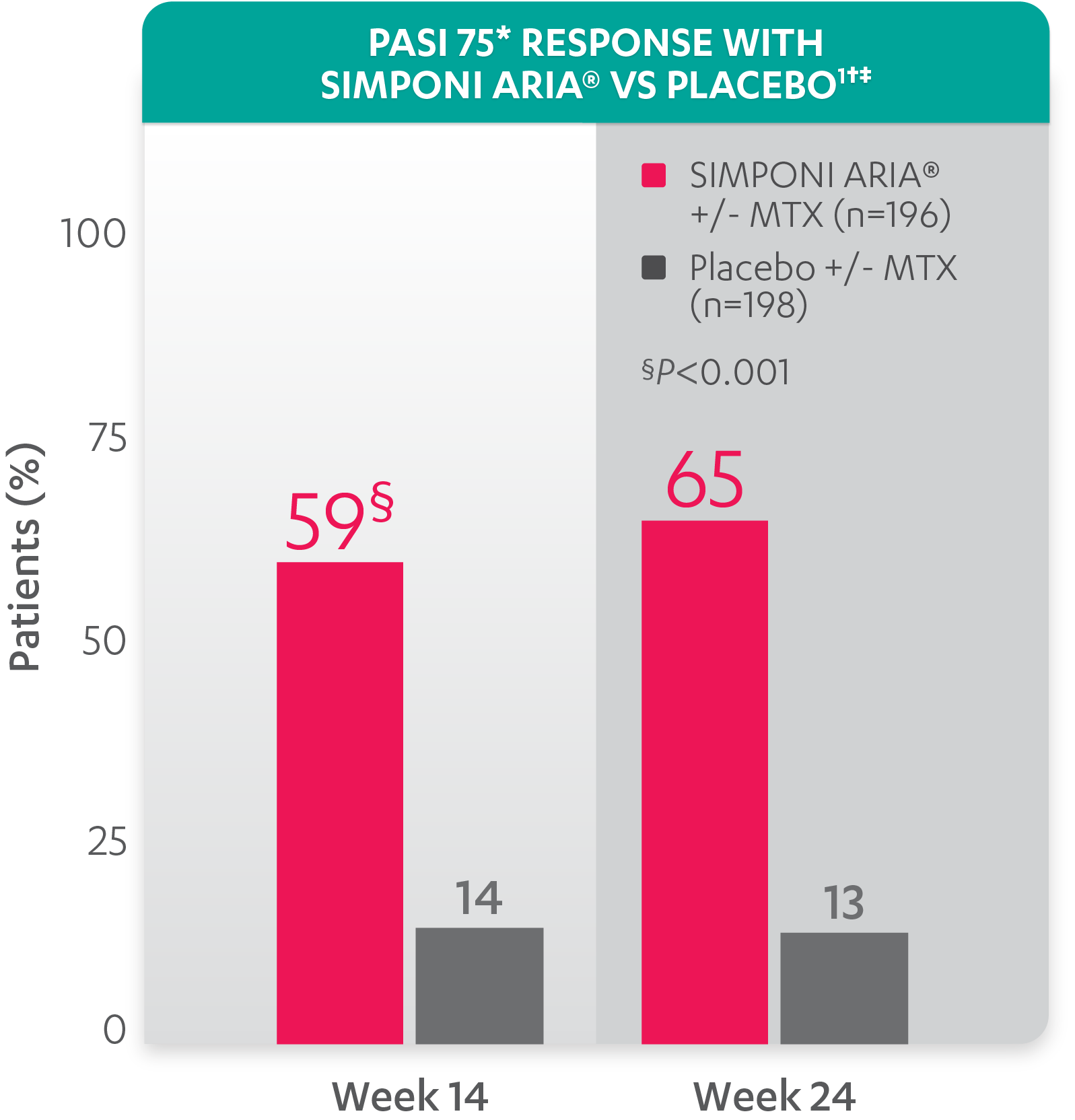
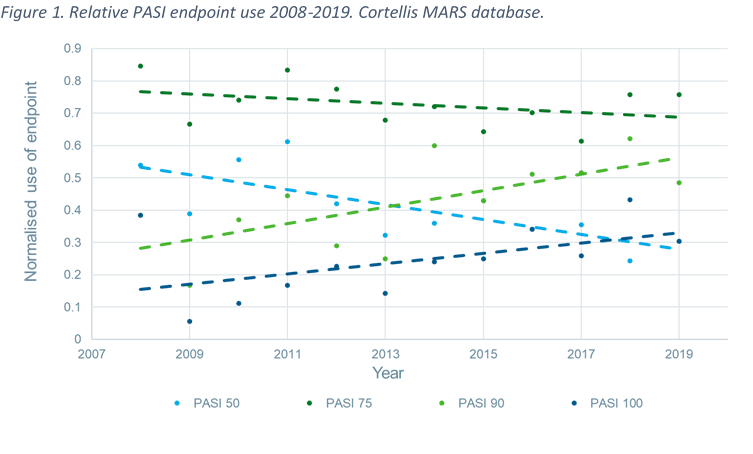
JCM | Free Full-Text | Use of Guselkumab for the Treatment of Moderate-to-Severe Plaque Psoriasis: A 1 Year Real-Life Study
Real-world efficacy of biological agents in moderate-to-severe plaque psoriasis: An analysis of 75 patients in Taiwan | PLOS ONE

Figure 1 | Rapid Response of Biologic Treatments of Moderate-to-Severe Plaque Psoriasis: A Comprehensive Investigation Using Bayesian and Frequentist Network Meta-analyses | SpringerLink

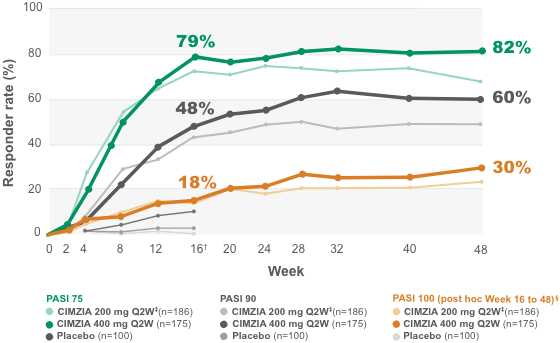
Secukinumab demonstrates high efficacy and a favorable safety profile over 52 weeks in Chinese patients with moderate to severe plaque psoriasis | Chinese Medical Journal
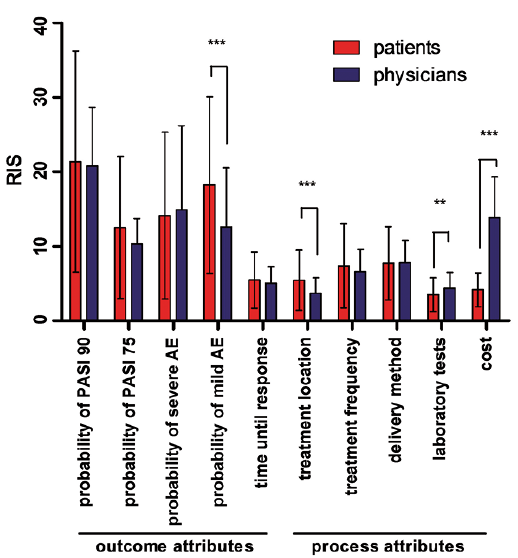
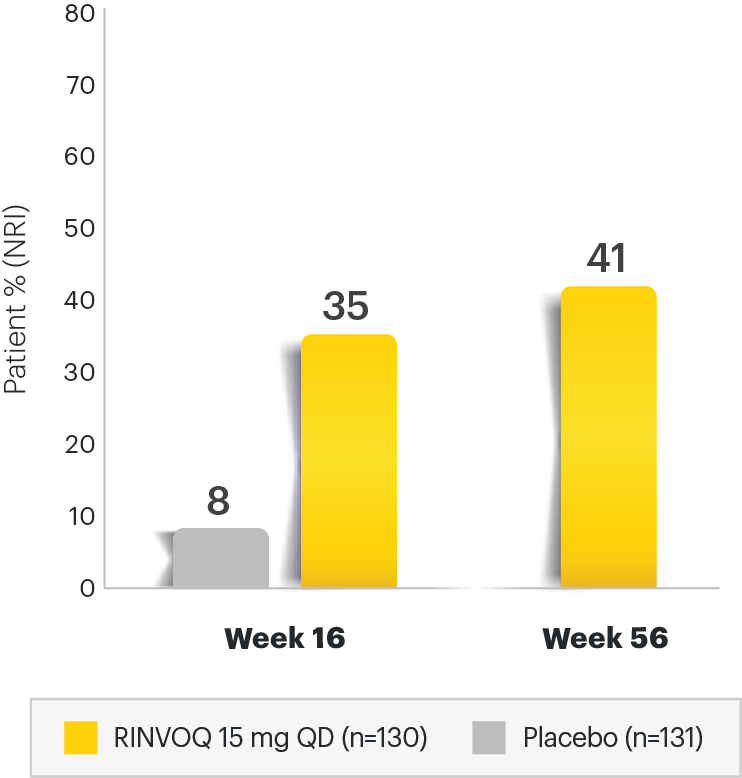
Patients' and Physicians' Preferences for Systemic Psoriasis Treatments: A Nationwide Comparative Discrete Choice Experiment (PsoCompare) | HTML | Acta Dermato-Venereologica
Secukinumab is superior to ustekinumab in clearing skin of subjects with moderate to severe plaque psoriasis: CLEAR, a randomize

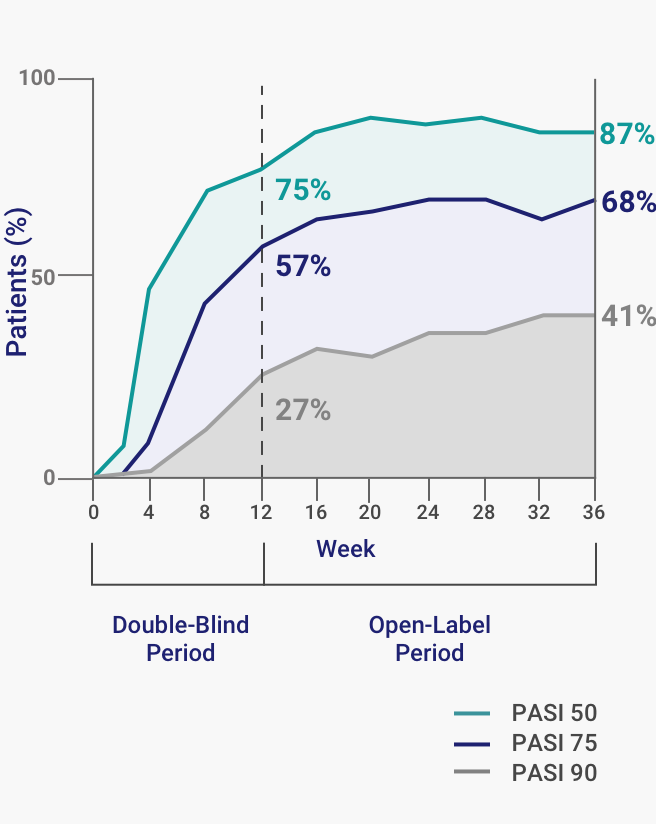
Long-term efficacy and safety of ixekizumab: A 5-year analysis of the UNCOVER-3 randomized controlled trial - ScienceDirect

A 50% reduction in the Psoriasis Area and Severity Index (PASI 50) is a clinically significant endpoint in the assessment of psoriasis - ScienceDirect
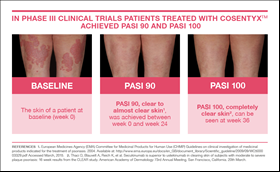
New Novartis data shows Cosentyx™ is significantly superior to Stelara® and clears skin (PASI 90) in nearly 80% of psoriasis patients | Novartis

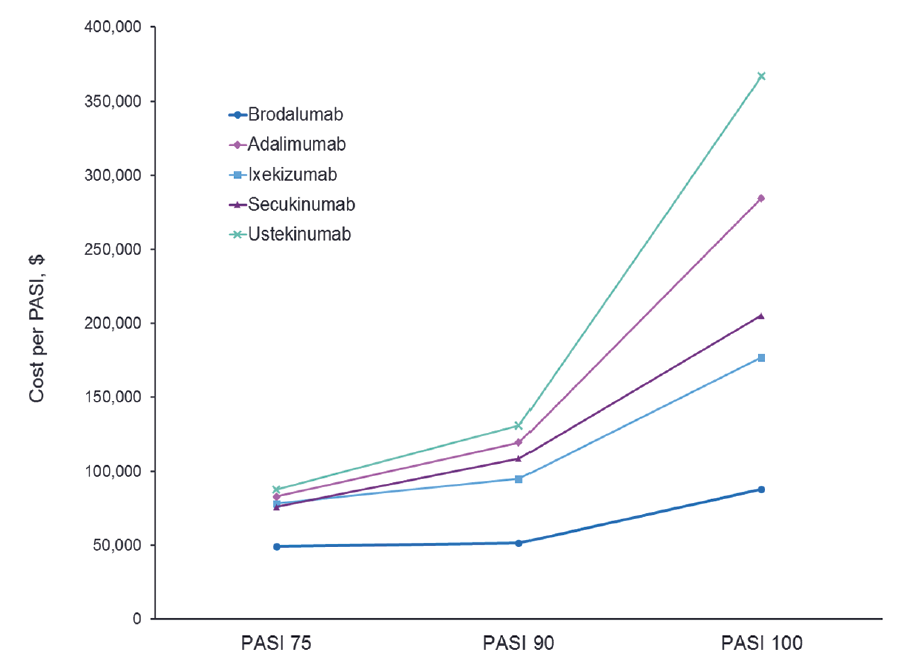
PASI 90 / 100 , DLQI 0 / 1 , and IL-17 Receptor / Cytokine : Does it Make a Difference and Are We Ambitious Enough ? | Semantic Scholar