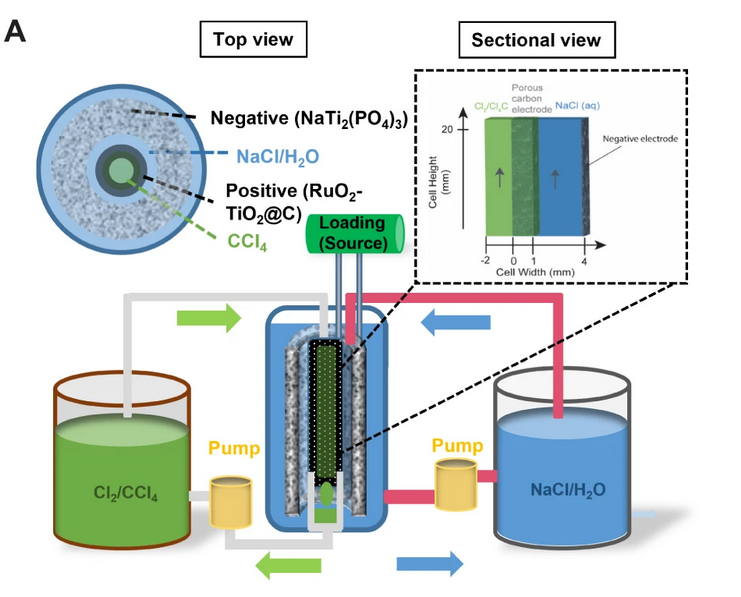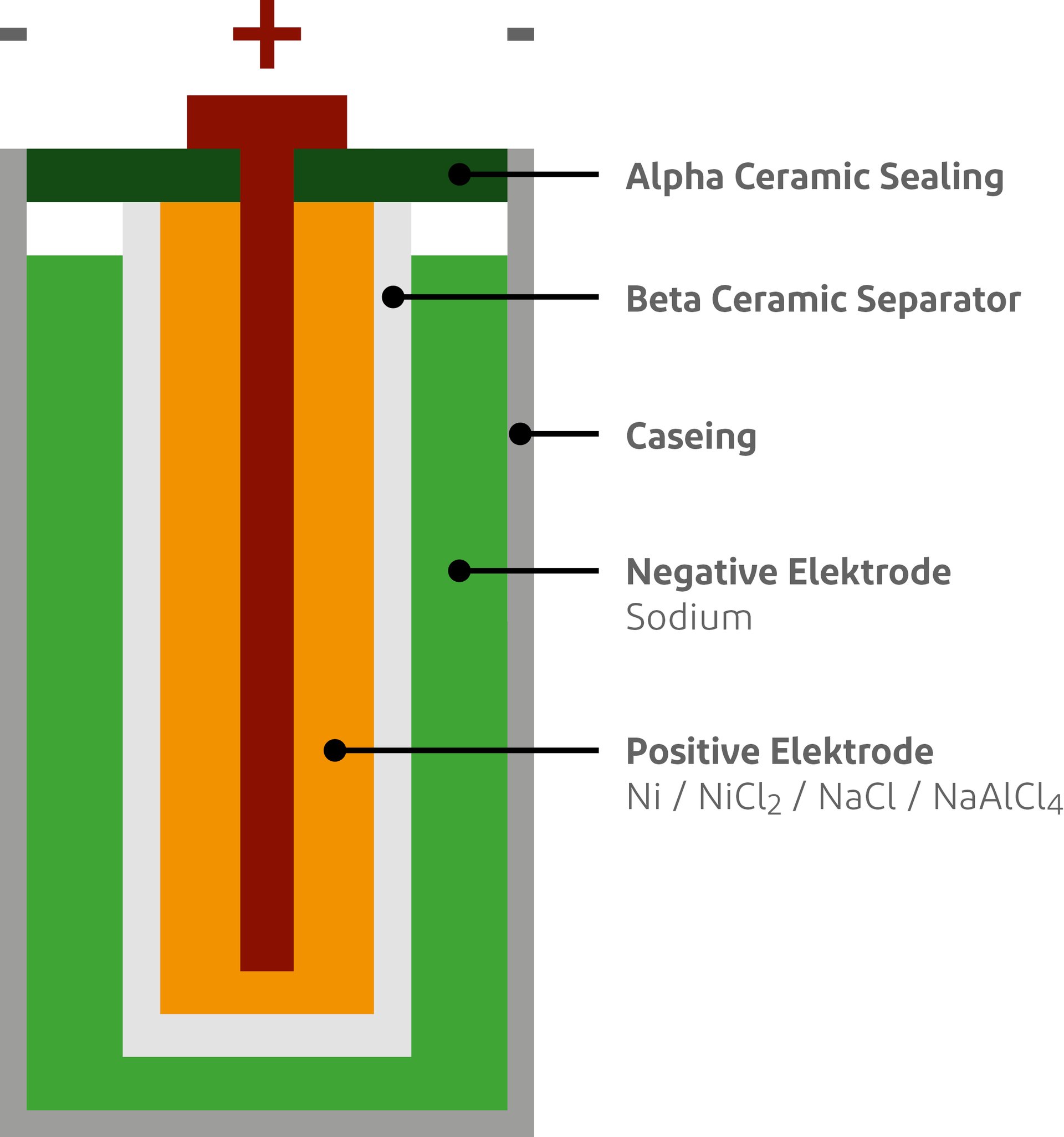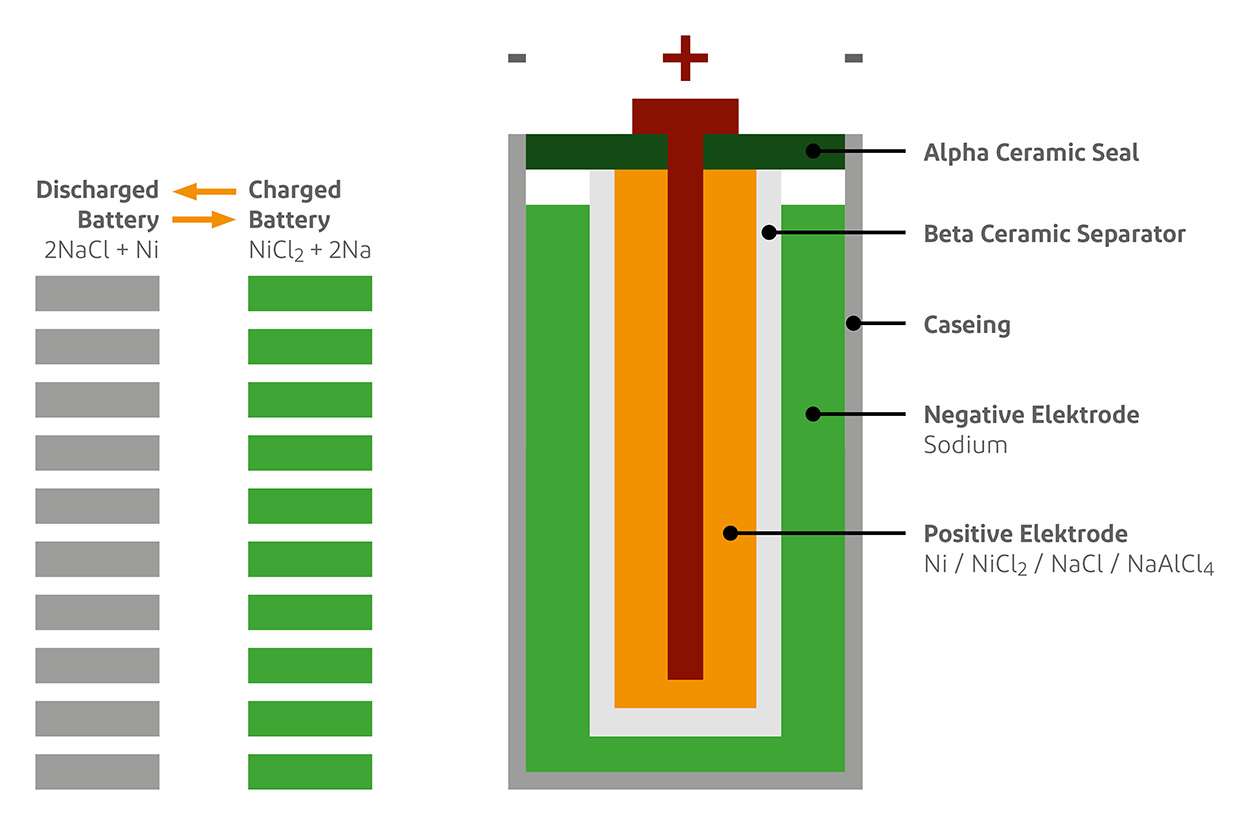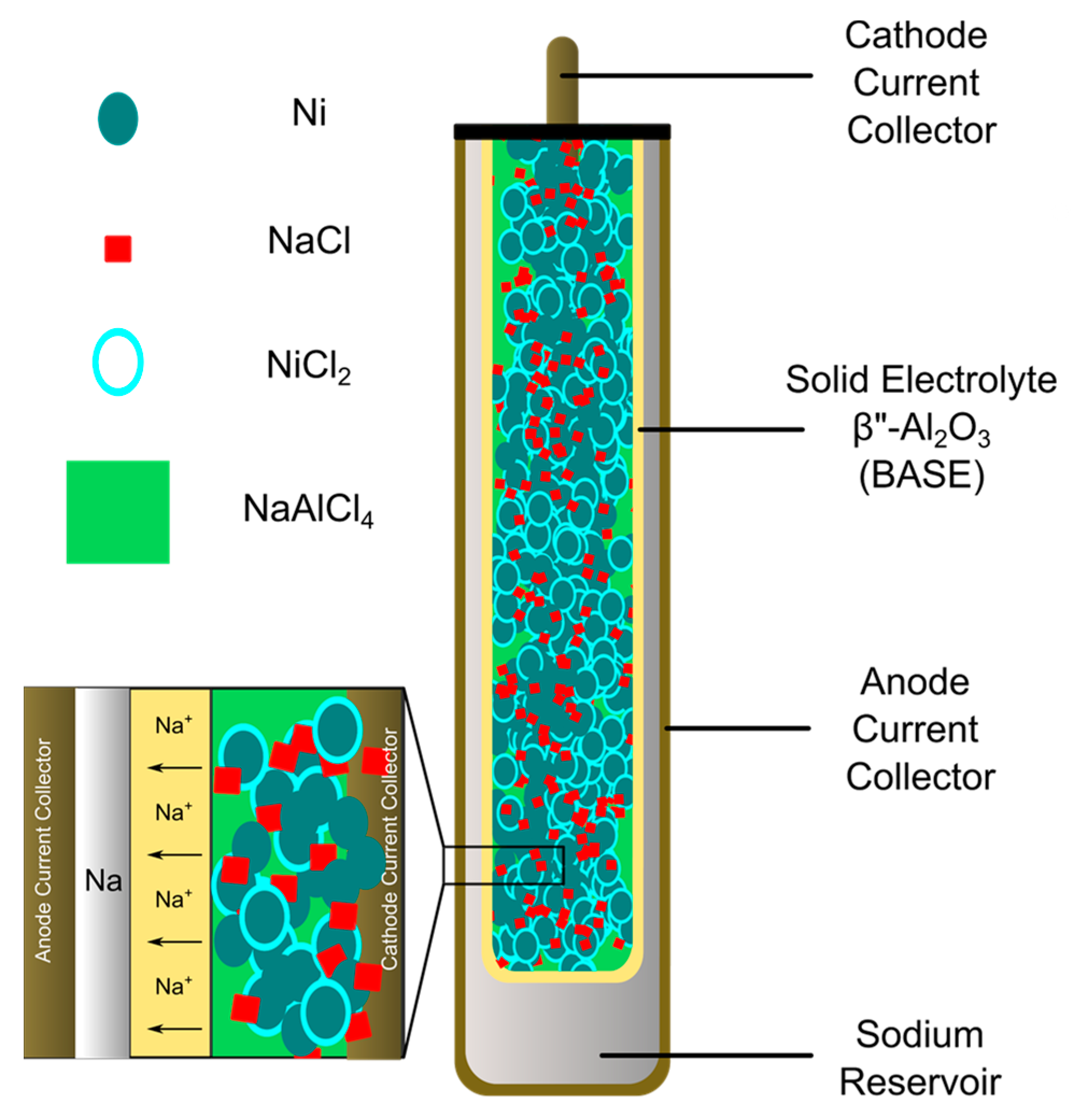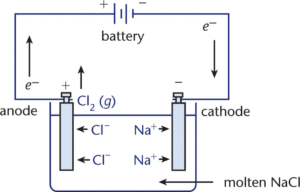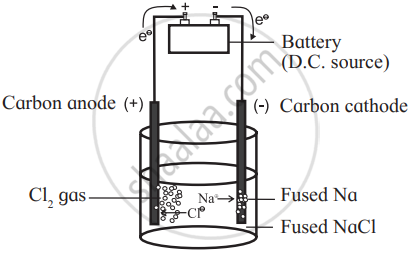
Explain construction, working in terms of cell reactions and the results of electrolysis of fused NaCl. - Chemistry | Shaalaa.com
![Sodium Nickel Chloride Battery [62] 4.1.3.2.3 Flow battery -Vanadium... | Download Scientific Diagram Sodium Nickel Chloride Battery [62] 4.1.3.2.3 Flow battery -Vanadium... | Download Scientific Diagram](https://www.researchgate.net/publication/305474151/figure/fig12/AS:837090497536010@1576589335322/Sodium-Nickel-Chloride-Battery-62-41323-Flow-battery-Vanadium-Redox-Battery-VRB.jpg)
Sodium Nickel Chloride Battery [62] 4.1.3.2.3 Flow battery -Vanadium... | Download Scientific Diagram

electrolysis of sodium chloride solution brine product equations electrodes anode cathode apparatus electrolyte cell sodium bromide potassium iodide gcse chemistry KS4 science igcse O level revision notes
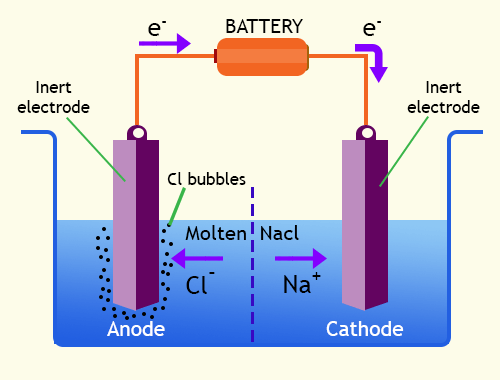
Electrolytic cells structure & importance, the difference between Electrolytic cell & Galvanic cell | Science online

Electrolysis of Molten NaCl (in Urdu/Hindi) - Oxidation and Reduction Reactions in Electrolytic Cell - YouTube

Long-Term Stable Hollowed Silicon for Li-Ion Batteries Based on an Improved Low-Temperature Molten Salt Strategy | ACS Omega

Sodium and Sodium‐Ion Batteries: 50 Years of Research - Delmas - 2018 - Advanced Energy Materials - Wiley Online Library

Electrochemically Induced Metallization of NaCl: Use of the Main Component of Salt as a Cost-Effective Electrode Material for Sodium-Ion Batteries | ACS Energy Letters
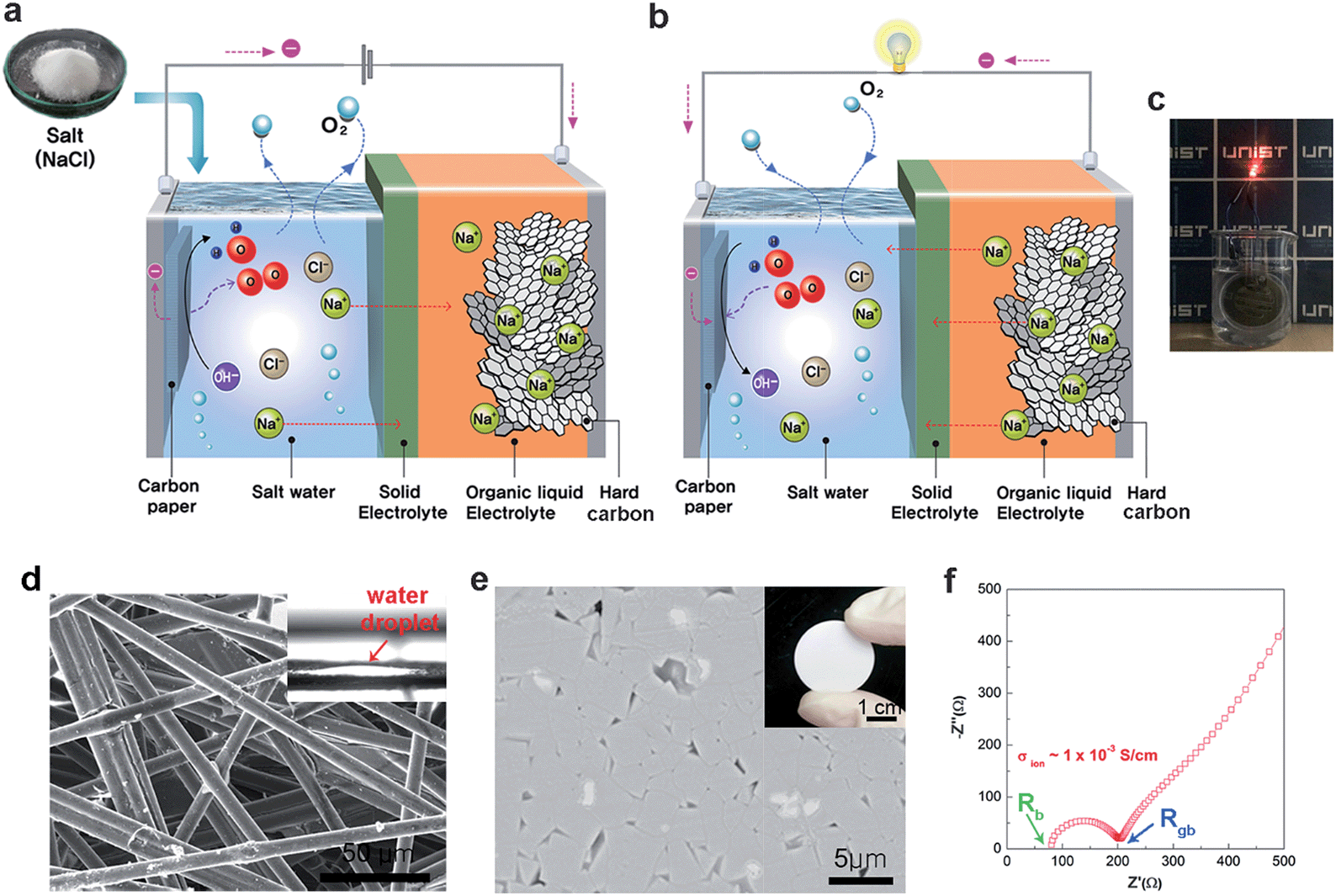
Saltwater as the energy source for low-cost, safe rechargeable batteries - Journal of Materials Chemistry A (RSC Publishing) DOI:10.1039/C6TA01274D